 
In the VSEPR model, thats covalent bonds and lone pairs. The electron domain geometry of NH3 is trigonal pyramidal, meaning that there are three electron domains around the central atom and that the shape of the. Moore, John W., Stanitski, Conrad L., Wood, James L., Kotz, John C., The Chemical World, Concepts and Applications, Second Edition, Volume 1, Harcourt Brace & Co., 1998, pages 397-447. Electron domains are the most probable area for finding an electron. Ebbing, Darrell D., Wrighton, Mark S., General Chemistry, Second Edition, Houghton Mifflin Co., 1987 pages 286-321.Ģ. To predict the molecular geometry select from the table below the 3D arrangement that has the same number of bond domainsand lone pairs of electrons.ġ. Determine the number of bond domains and the number of lone pairs of electrons.ģ. Halogens and noble gases can expand their octet.Ģ. Note: To chose the central atom as the one with the smallest number of valence electrons or if they all have the same number of valence electrons then choose the one in the least amount. The primary difference is that with molecular geometry we include only the atoms in naming the. Draw Lewis structures for the molecular formula given. An explanation of the difference between molecular geometry and electron geometry. How to use the table to predict molecular geometry.ġ. For example, the molecule ethylene, H 2C=CH 2, has the carbon-carbon atoms sharing four electrons but one bond domain between the two carbon atoms. In any case, if two atoms share two electrons or more they will have one bond domain. For ease of understanding we will call a bond domain the electrons that hold together two atoms. The following table will help you understand how molecular geometry can be predicted using the VSPER model. As such, this model of molecular geometry is often referred to as the valence shell electron pair repulsion (VSEPR) theory. On the other hand it takes into account the very important Pauli exclusion principle where each electron pair must occupy a different spatial region about an atom. We conclude that molecular geometry is determined by minimizing the mutual repulsion of the valence shell electron pairs. 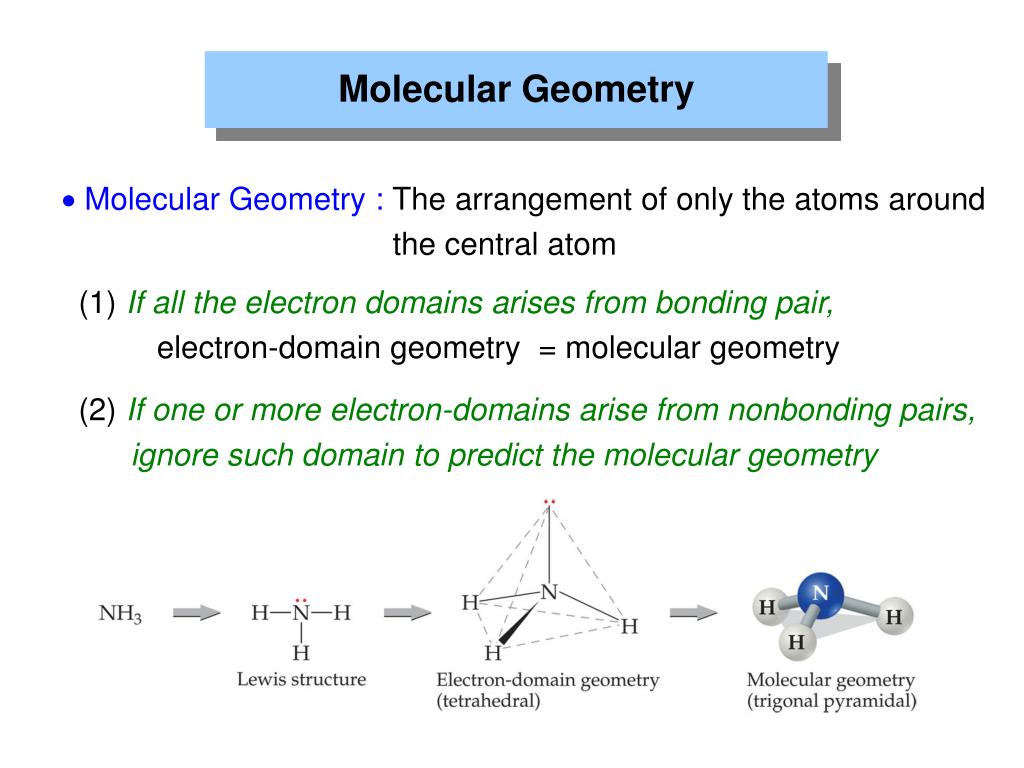 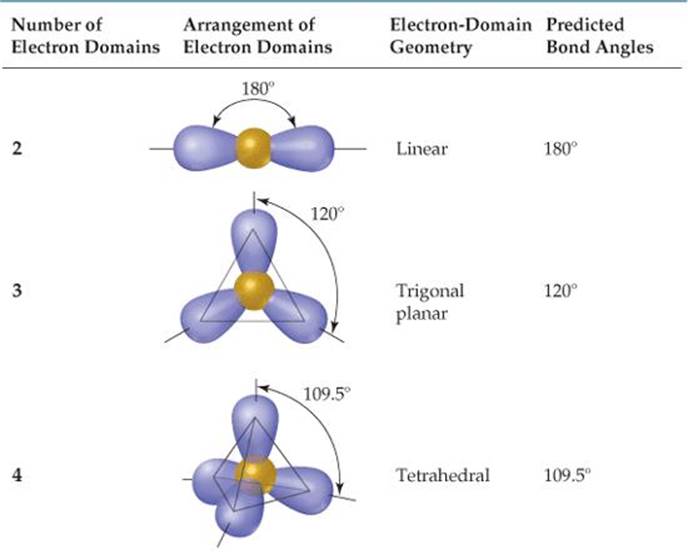
On the first hand it minimizes repulsion between electrons due to electrostatic interactions. The VSPER model is based on two important principles. In contrast, molecular geometry focuses solely on the arrangement of atoms in the molecule. This model proposes that electrons are arranged around atoms in pairs such that they are kept as far away as possible. Key Takeaways Electron geometry describes the spatial arrangement of electron pairs (including bonding and lone pairs) around a central atom in a molecule. The valence shell electron pair repulsion (VSPER pronounced "vesper") model provides some useful tools for predicting molecular geometries. Atoms have a definite three-dimensional spacearrangement relative to each other in a molecule. can also be observed in the microscopic world. The geometrical arrangements seen in nature, i.e., flowers,stones, trees, etc. The electron-domain geometry indicated by VSEPR describes the arrangement of all bonding and nonbonding electron domains. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
